- L’utilisation de la combinaison Aspirine + Inhibiteur de P2Y12 (DAPT pour ‘Dual antiplatelet therapy) est devenu le traitement de référence pour la prise en charge des patients après un événement cardiovasculaire majeur mais un nouvel événement cardiovasculaire survient chez 10% des patients au cours du suivi
- La thérapie antithrombotique triple avec DAPT + rivaroxaban, un inhibiteur du facteur Xa, permet de réduire les événements cardiovasculaires, mais avec un taux significativement plus élevé de complications de saignement
- Des études ont suggéré que la combinaison rivaroxaban + Inhibiteur de P2Y12 a une efficacité similaire à la combinaison DAPT classique, mais avec un risque de saignement plus faible
L’étude GEMINI-ACS-1 a été entreprise pour comparer les 2 types de combinaisons thérapeutiques: stratégie antithrombotique double (antiplaquettaire et anticoagulant) par rapport à stratégie antiplaquettaire double (aspirine et inhibiteur de P2Y12). Les résultats de cette étude, publiés dans The Lancet (Ohman EM et al. 2017), indiquent que le risque hémorragique est identique dans les 2 groupes de traitement. L’abandon de l’aspirine lorsqu’on utilise une stratégie combinant inhibiteur de P2Y12 et faible dose d’anticoagulant permet donc d’avoir un risque hémorragique pas plus élevé q’une bithérapie antiplaquettaire conventionnelle. L’efficacité thérapeutique comparée sur le risque ischémique de ces 2 stratégies nécessitera de nouvelles études.
Ohman, E. M., Roe, M. T., Steg, P. G., James, S. K., Povsic, T. J., White, J., Rockhold, F., Plotnikov, A., Mundl, H., Strony, J., Sun, X., Husted, S., Tendera, M., Montalescot, G., Bahit, M. C., Ardissino, D., Bueno, H., Claeys, M. J., Nicolau, J. C., Cornel, J. H., Goto, S., Kiss, R. G., Guray, U., Park, D. W., Bode, C., Welsh, R. C. and Gibson, C. M.
Clinically significant bleeding with low-dose rivaroxaban versus aspirin, in addition to P2Y12 inhibition, in acute coronary syndromes (GEMINI-ACS-1): a double-blind, multicentre, randomised trial
Lancet 2017. March. PubMed.
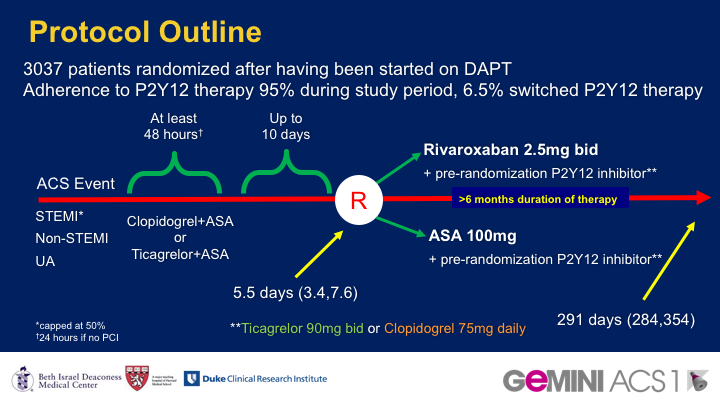
BACKGROUND: Dual antiplatelet therapy (DAPT), aspirin plus a P2Y12 inhibitor, is the standard antithrombotic treatment following acute coronary syndromes. The factor Xa inhibitor rivaroxaban reduced mortality and ischaemic events when added to DAPT, but caused increased bleeding. The safety of a dual pathway antithrombotic therapy approach combining low-dose rivaroxaban (in place of aspirin) with a P2Y12 inhibitor has not been assesssed in acute coronary syndromes. We aimed to assess rivaroxaban 2.5 mg twice daily versus aspirin 100 mg daily, in addition to clopidogrel or ticagrelor (chosen at investigator discretion before randomisation), for patients with acute coronary syndromes started within 10 days after presentation and continued for 6-12 months.
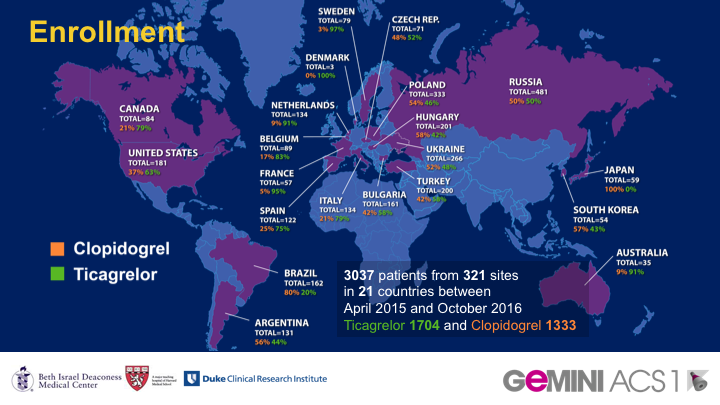
METHODS: In this double-blind, multicentre, randomised trial (GEMINI-ACS-1) done at 371 clinical centres in 21 countries, eligible patients were older than 18 years with unstable angina, non-ST segment elevation myocardial infarction (NSTEMI) or ST segment elevation myocardial infarction (STEMI), with positive cardiac biomarkers and either ischaemic electrocardiographic changes or an atherosclerotic culprit lesion identified during angiography. Participants were randomly assigned (1:1) within 10 days after admission for the index acute coronary syndromes event to either aspirin or rivaroxaban based on a computer-generated randomisation schedule. Randomisation was balanced by using randomly permuted blocks with size of four and was stratified based on the background P2Y12 inhibitor (clopidogrel or ticagrelor) intended to be used at the time of randomisation. Investigators and patients were masked to treatment assignment. Patients received a minimum of 180 days of double-blind treatment with rivaroxaban 2.5 mg twice daily or aspirin 100 mg daily. The choice of clopidogrel or ticagrelor during trial conduct was not randomised and was based on investigator preference. The primary endpoint was thrombolysis in myocardial infarction (TIMI) clinically significant bleeding not related to coronary artery bypass grafting (CABG; major, minor, or requiring medical attention) up to day 390. Primary analysis was by intention to treat. This study is registered with ClinicalTrials.gov, number NCT02293395.
FINDINGS: Between April 22, 2015, and Oct 14, 2016, 3037 patients with acute coronary syndromes were randomly assigned; 1518 to receive aspirin and 1519 to receive rivaroxaban. 1704 patients (56%) were in the ticagrelor and 1333 (44%) in the clopidogrel strata. Median duration of treatment was 291 days (IQR 239-354). TIMI non-CABG clinically significant bleeding was similar with rivaroxaban versus aspirin therapy (total 154 patients [5%]; 80 participants [5%] of 1519 vs 74 participants [5%] of 1518; HR 1.09 [95% CI 0.80-1.50]; p=0.5840).
INTERPRETATION: A dual pathway antithrombotic therapy approach combining low-dose rivaroxaban with a P2Y12 inhibitor for the treatment of patients with acute coronary syndromes had similar risk of clinically significant bleeding as aspirin and a P2Y12 inhibitor. A larger, adequately powered trial would be required to definitively assess the efficacy and safety of this approach.
FUNDING: Janssen Research & Development and Bayer AG.